Onion maggot and seedcorn maggot (Delia antiqua and Delia platura)
Vegetable IPM Fact Sheet
The onion maggot (Delia antiqua; OM) and seedcorn maggot (Delia platura; SCM) (Diptera: Anthomyiidae) are the most problematic insect pests in onions early in the season. Heavy infestations can reduce plant stands by up to 100% if not controlled. Effective management relies on understanding their identification, life cycle, and control options.
In this fact sheet
- Identification of onion maggot and seedcorn maggot
- Pest status of onion maggot and seedcorn maggot
- Life cycle of onion maggot and seedcorn maggot
- Management of onion maggot and seedcorn maggot

Identification of onion maggot and seedcorn maggot
Adults of both species resemble house flies. In general, onion maggot flies are larger than seedcorn maggot flies. In the same way, onion maggot larvae achieve larger sizes than seedcorn maggot larvae. Distinguishing between onion maggot and seedcorn maggot can be challenging because they are closely related and have a similar appearance. However, subtle morphological differences exist in both adult and larval stages. Positive identification often requires microscopic examination, especially for larvae.
Onion maggot
- Eggs: Eggs are elongated, white, and 1.1–1.3 mm long. The surface has a coarse reticulated pattern, characterized by deep depressions bordered by relatively thick ridges.
- Larvae: Larvae are cream colored, generally 7.5–12.0 mm long, and have 8–12 (rarely 13 or 14) papillae in their anterior spiracle (Figure 1). Tubercle X is small and located ventrally to tubercles A and B; tubercle A is usually wider near the base than tubercle B, but widely separated apically.
- Pupae: Pupae are 4.5–7 mm long.
- Adults: Body length ranges from 5.0 to 7.2 mm, silvery grey in color, paler on the pleuron (thoracic segment of an insect between the tergum and sternum). The fore tibia usually has three or more bristles, and the hind tibia has seven to fifteen erect bristles (fewer is more common: Figures 2 and 3).
Seedcorn maggot
- Eggs: Eggs are elongated, white, and 0.97–1.05 mm long. The surface has a delicate reticulated pattern with shallow depressions separated by narrower edges.
- Larvae: Larvae are 5.9–6.8 mm long and have 6–8 papillae (rarely 9) in the anterior spiracle (Figure 1). Tubercle X, tubercle A, and tubercle B present.
- Pupae: Pupae are 4.3–5.1 mm long.
- Adults: Adults are 2.9–5.8 mm long. Fore tibia usually with two and rarely three bristles, mid tibia usually with less than five bristles (rarely six), and hind tibia with more than eighteen short erect bristles. Hind femur with no more than 6 preapical posteroventral bristles restricted to the apical fifth of the surface (Figures 2 and 3).
The seedcorn maggot and the bean seed maggot (Delia florilega [Zetterstedt, 1845]) are morphologically indistinguishable during their egg, larval, and pupal stages. However, adult bean seed maggots are 2.8–4.8 mm long and are typically smaller than seedcorn maggot adults. Bean seed maggot adults also possess only one posteroventral bristle, compared with two or three on the seedcorn maggot. Sometimes these bristles might be lost in the seedcorn maggot.
Additionally, male bean seed maggot adults can be differentiated from male seedcorn maggot adults by a line of long setulae on the first tarsomere of their midleg. In contrast, male seedcorn maggot adults have only short setulae on this same segment.
Figure 1. Onion maggot and seedcorn maggot have 8 papillae and 6 papillae within the anterior spiracle, respectively.
Picture credit: Nathan Hesler.
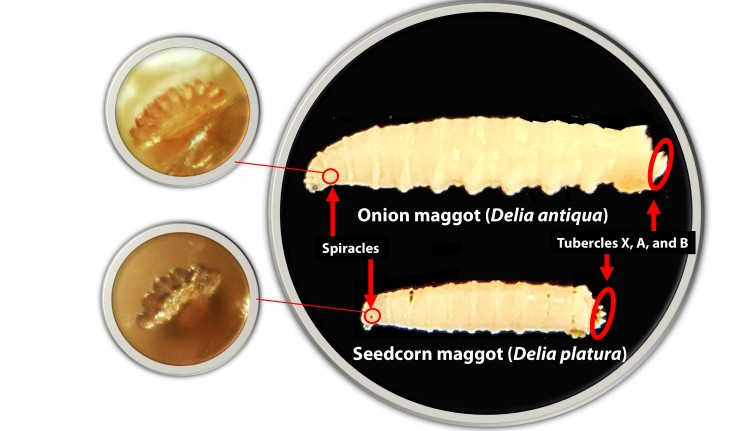

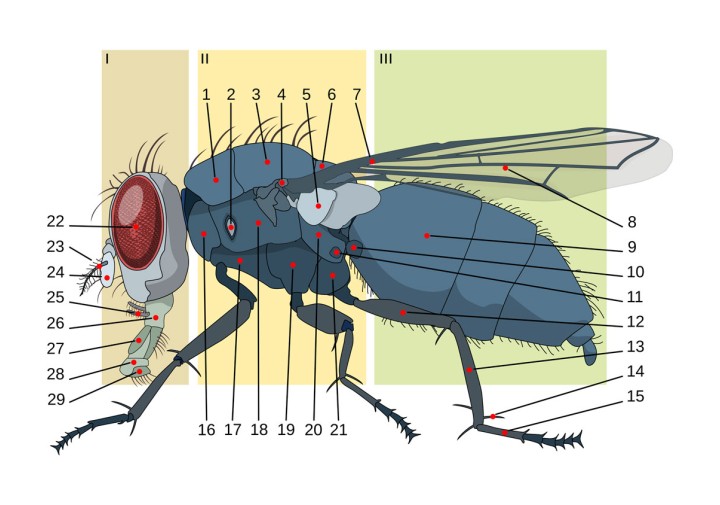
Figure 2 (left). Diagram of an onion maggot adult.
Picture credit: Joe Ogrodnick.
Figure 3 (right). Schematic of fly anatomy.
Picture Credit: Al2 and derivative work by: Giancarlodessi and Georg-Johann (CC BY 3.0).
Figure 3 labels:
I: head; II: thorax III: abdomen. — 1: prescutum 2: anterior spiracle 3: scutum 4: basicosta 5: calypters 6: scutellum 7: wing vein 8: wing 9: abdominal segment 10: haltere 11: posterior spiracle 12: femur 13: tibia 14: spur 15: tarsus 16: propleuron 17: prosternum 18: mesopleuron 19: mesosternum 20: metapleuron 21: metasternum 22: compound eye 23: arista 24: antenna 25: maxilary palps 26: labium 27: labellum 28: pseudotracheae 29: tip.
Pest status of onion maggot and seedcorn maggot
The onion maggot and the seedcorn maggot have different life histories. The onion maggot is a specialist that feeds only on plants in the Allium genus like onion (A. cepa), garlic (A. sativum), leek (A. ampeloprasum), and chives (A. schoenoprasum). Leek, chive, and garlic chive are less preferred by onion maggot than onion.
In contrast, the seedcorn maggot is a generalist that feeds on a wide variety of crops such as beans (Phaseolus spp.), peas (Pisum spp.), corn (Zea mays), soybean (Glycine max), cucumber (Cucumis sativus), melon (Cucumis melo), potato (Solanum tuberosum), and all of the crops listed above in the Allium genus. Both pests attack allium crops throughout the northern onion production areas in the US and across the world, but differ in abundance and pest status in each region. Larvae feed on the below-ground parts of the onion plant, which can kill the plant or render it unmarketable (See Figures 4 through 9).
Life cycle of onion maggot and seedcorn maggot
Onion maggot (green diagram)
The onion maggot has three generations per year, and larvae from the third generation overwinter in the soil as pupae. Onion maggot flies emerge from pupae in late April through May, mate, and lay eggs in the soil at the base of onion plants. Onion seedlings are particularly susceptible to these first-generation larvae because plants are more vulnerable at a young stage. The onion maggot's life cycle can last 37 to more than 60 days, with three instars and a pupal stage.
First-generation onion maggot adults emerge from pupae around mid-May in the northeastern United States, with peak flights occurring around 712–1018 degree days using a base 40°F model (378–548-degree day base 4°C) [Figure 10].
Seedcorn maggot (yellow diagram)
The seedcorn maggot has three to five generations per year, but the earlier generations tend to be more numerous and problematic in the spring. The seedcorn maggot will lay eggs in decaying organic material. Seedcorn maggot's lifecycle is significantly shorter than the onion maggot’s, lasting from 19 to 36 days.
First-generation seedcorn maggot adults require an average accumulated degree days for spring emergence between 91 to 105 degree days at a base temperature of 3.9°C (163.8 to 189 degree days at a base temperature of 39.02°F) [Figure 10].


Figure 10. Life cycle of onion maggot (green) and seedcorn maggot (yellow). Picture credits: Joe Ogrodnick.
Management of onion maggot and seedcorn maggot in onions
There is no curative treatment for the onion maggot and seedcorn maggot because once damage occurs, nothing can be done; therefore, management relies on preventive tactics. Maggot management is achieved through chemical control and cultural practices.
Growers should implement preventative tactics for seedcorn and onion maggots when their field management practices create conditions favorable for these pests. For seedcorn maggots, the primary risk factor is the presence of freshly incorporated organic matter. Fields with recent applications of manure, green cover crops, or significant crop residue are highly attractive to egg-laying females and require proactive management. For onion maggots, preventative measures are necessary when infestations have been high in the previous years and when potential onion plant damage by equipment, cultivation, or disease occurs, as this stress attracts the flies.
Currently, maggots are managed principally with insecticides applied as either seed treatments or transplant tray drenches. Cultural practices include crop rotation and sanitation, delayed planting, biological control, and sterile insect release. Some of these management practices are not economically practical in commercial-scale onion production but could be effective on small diversified farms.
Host plant resistance
- No resistant cultivars to maggots exist.
Biological control
- Beetles.
Around 100 species of beetles in the rove beetle (Staphylinidae) and ground beetle (Carabidae) families are generalist predators of eggs and larvae of maggots. Because these beetles are not specialist predators, they have not been successfully integrated into Delia spp. management plans. Not commercially available (Figure 13). - Parasitoid wasps.
Aphaereta pallipes will parasitize Delia spp maggots in the laboratory, but has not been detected in the field. Not commercially available. - Fungi.
Three groups of fungi (Metarhizium spp., Beauveria bassiana, and Entomophthora muscae) are responsible for infecting and killing maggots in onions (Figure 12). However, fungal isolates tend to cause less mortality of larvae than adults. All are commercially available. Note: An antagonistic effect is likely between entomopathogenic fungi and fungicides used to manage onion plant diseases. - Nematodes.
Steinernema feltiae and Heterorhabditis bacteriophora can infect Delia spp. Nematodes are commercially available. Some species may be compatible with soil-applied insecticides, but others will not. Soil conditions likely impact performance of nematodes.
Cultural control
- Crop rotation.
This practice can reduce risk of damage by maggots, but its success will be dependent on distance from overwintering sites (need to be greater than 1 mile–1.6 km). - Sanitation.
- Refers to practices that reduce pest problems by removing host resources for the pest. Removing bulbs left behind after harvest from the previous season can be helpful. “Volunteer” onion plants, which grow from bulbs left in the field the previous year, can serve as a host for larvae at the beginning of the season.
- Seedcorn maggot females prefer to lay eggs in soils rich in decaying organic matter, such as cover crops, green manure, or crop residues. High organic matter levels provide suitable sites and food sources for larvae, often increasing maggot populations and damage risk. Managing organic matter through crop rotation and sanitation can help reduce maggot infestations.
- Delayed planting.
- This practice disrupts the synchrony between the first generation of onion maggot and the crop, which can allow the crop to escape the pest in time. Additionally, flies prefer to oviposit on larger, earlier planted onion plants, rather than on smaller, later planted ones. A drawback of this strategy is that late plantings of onions may either generate smaller bulbs or no bulbs, which is determined physiologically.
- Transplanting onions can have a lower risk of damage than seeded ones because transplanted onions are often larger and more resilient to attack compared with smaller, seeded onion plants.
Behavioral control
Sterile insect technique is a management tactic in which males are reared and sterilized via radiation and then released into onion fields to mate with females. Irradiated males compete with non-irradiated males for mating opportunities with fertile females, which reduces the number of viable offspring. This strategy has been effectively utilized for onion maggot management in the Netherlands and Quebec, but is not widely adopted due to cost and labor concerns.
Chemical control
- Control of the maggot complex (Diptera: Anthomyiidae) in onions is mainly achieved using seed treatments and transplant tray drenches in transplanted onions (Figure 11).
- Foliar sprays have been used to target adult flies before oviposition, but this tactic is not effective because flies disperse and avoid insecticide exposure.
- To control maggots, a range of insecticide seed treatments can be used. These include spinosad (IRAC Group 5), cyromazine (IRAC Group 17), and three neonicotinoids: thiamethoxam, clothianidin, and imidacloprid (all belonging to IRAC Group 4A)[Figure 14].
- Consult current pest management guidelines (i.e., Cornell integrated crop and pest management guidelines for vegetable crops: https://cropandpestguides.cce.cornell.edu) for the most effective options.
- Additionally, the efficacy of an insecticide seed treatment may differ depending on the prevalence of onion maggot and seedcorn maggot in a particular region.
- For resistance management, consider rotating insecticide seed treatments among IRAC groups every year.
Any time you use a pesticide, you must read and follow the label directions and comply with all applicable laws and regulations related to pesticide use. Also, be sure that any pesticide used is approved for use in your country and state/province.
Figure 14. Comparison of onion plants not treated with insecticides (left side of photo) and plants protected from maggots using insecticide seed treatments (right side of photo).
Picture Credit: Brian Nault.

Authors
Leonardo D. Salgado
Graduate Research Assistant, Department of Entomology, Cornell AgriTechBrian A. Nault
Professor, Department of Entomology, Cornell AgriTech
Last updated: June 2025
Cornell University’s College of Agriculture and Life Sciences Extension and Outreach Assistantship program supported graduate student Leonardo Salgado in his position as an Extension and Outreach Assistant in Spring 2025. Authors are also thankful to Marion Zuefle and Henry Zelenak of Cornell Integrated Pest Management for reviewing and creating the online version of this fact sheet, respectively.
- Carruthers RI, Haynes DL. 1985. Laboratory transmission and in vivo incubation of Entomophthora muscae (Entomophthorales: Entomophthoracae) in the onion fly, Delia antiqua (Diptera: Anthomyiidae). Journal of Invertebrate Pathology. 45(3):282–287. https://doi.org/10.1016/0022-2011(85)90105-3
- Davidson G, Chandler D. 2005. Laboratory evaluation of entomopathogenic fungi against larvae and adults of onion maggot (Diptera: Anthomyiidae). Journal of Economic Entomology. 98(6):1848–1855. https://doi.org/10.1093/jee/98.6.1848
- Filgueiras CC, Shields EJ, Nault BA, et al. 2023. Entomopathogenic nematodes for field control of onion maggot (Delia antiqua) and compatibility with seed treatments. Insects. 14(7):623. https://doi.org/10.3390/insects14070623
- Funderburk JE, Higley LG, Pedigo LP. 1984. Seedcorn maggot (Diptera: Anthomyiidae) phenology in central Iowa and examination of a thermal-unit system to predict development under field conditions. Environmental Entomology. 13(1):105–109. https://doi.org/10.1093/ee/13.1.105
- Higley LG, Pedigo LP. 1984. Seedcorn maggot (Diptera: Anthomyiidae) population biology and aestivation in central Iowa. Environmental Entomology. 13(5):1436–1442. https://doi.org/10.1093/ee/13.5.1436
- Loosjes M. 1976. Ecology and genetic control of the onion fly, Delia antiqua (Meigen). Wageningen: PUDOC (Agricultural research reports).
- McFerson JR, Walters TW, Eckenrode CJ. 1996. Variation in Allium spp. damage by onion maggot. HortSci. 31(7):1219–1222. https://doi.org/10.21273/HORTSCI.31.7.1219
- Miles M. 1955. Studies of British anthomyiid flies. VII.—The Onion-Fly Complex. Bulletin of Entomological Research. 46(1):21–26. https://doi.org/10.1017/S000748530003073X
- Miles M. 1958. Studies of British anthomyiid flies. IX.—Biology of the Onion Fly, Delia antiqua (Mg.). Bulletin of Entomological Research. 49(2):405–414. https://doi.org/10.1017/S0007485300053700
- Nault BA, Straub RW, Taylor AG. 2006. Performance of novel insecticide seed treatments for managing onion maggot (Diptera: Anthomyiidae) in onion fields. Crop Protection. 25(1):58–65. https://doi.org/10.1016/j.cropro.2005.03.020
- Olaya-Arenas P, Cho CY-L, Olmstead D, et al. 2024. Degree-day models for predicting adult Delia platura (Diptera: Anthomyiidae) spring flight and first emergence in New York State. Musser F, editor. Journal of Economic Entomology. 117(5):2181–2185. https://doi.org/10.1093/jee/toae148
- Salgado LD, Nault BA. 2023. Evaluating foliar insecticide applications and seed treatments for onion maggot control in onion, 2022. Arthropod Management Tests. 48(1):tsad009. https://doi.org/10.1093/amt/tsad009
- Salgado LD, Wilson RG, Nault. B. A. 2024. Maggots-Be-Gone an update on insecticide seed treatment performance. Onion World: 6–9. Available on https://issuu.com/columbiamediagroup/docs/onion_world_december_2024/6
- Savage J, Fortier A-M, Fournier F, et al. 2016. Identification of Delia pest species (Diptera: Anthomyiidae) in cultivated crucifers and other vegetable crops in Canada. Canadian Journal of Arthropod Identification. 29. https://doi.org/10.3752/cjai.2016.29
- Throne JE, Eckenrode CJ. 1985. Emergence patterns of the seedcorn maggot, Delia platura (Diptera: Anthomyiidae). Environmental Entomology. 14(2):182–186. https://doi.org/10.1093/ee/14.2.182
- Walters TW, Eckenrode CJ. 1996. Integrated Management of the onion maggot (Diptera: Anthomyiidae). Journal of Economic Entomology. 89(6):1582–1586. https://doi.org/10.1093/jee/89.6.1582
- Whistlecraft JW, Harris CR, Tolman JH, et al. 1985. Mass-rearing Technique for Aleochara bilineata (Coleoptera: Staphylinidae). Journal of Economic Entomology. 78(4):995–997. https://doi.org/10.1093/jee/78.4.995
- Whistlecraft JW, Harris CR, Tomlin AD, et al. 1984. Mass rearing technique for a braconid parasite, Aphaereta pallipes (Say) (Hymenoptera: Braconidae). Journal of Economic Entomology. 77(3):814–816. https://doi.org/10.1093/jee/77.3.814
- Wilson RG, Orloff SB, Taylor AG. 2015. Evaluation of insecticides and application methods to protect onions from onion maggot, Delia antiqua, and seedcorn maggot, Delia platura, damage. Crop Protection. 67:102–108. https://doi.org/10.1016/j.cropro.2014.10.002
- Yildrim E, Hoy CW. 2003. Interaction between cyromazine and the entomopathogenic nematode Heterorhabditis bacteriophora Poinar “GPS11” for control of onion maggot, Delia antiqua (Meigen). Crop Protection. 22(7):923–927. https://doi.org/10.1016/S0261-2194(03)00091-7
- Zhang H, Wu S, Xing Z, et al. 2016. Bioassay and scanning electron microscopic observations reveal high virulence of entomopathogenic fungus, Beauveria bassiana, on the onion maggot (Diptera: Anthomyiidae) adults. Journal of Economic Entomology. 109(6):2309–2316. https://doi.org/10.1093/jee/tow235
- Figure 1. Onion maggot and seedcorn maggot have 8 papillae and 6 papillae within the anterior spiracle, respectively. Picture credit: Nathan Hesler.
- Figure 2. Diagram of an onion maggot adult. Picture credit: Joe Ogrodnick.
- Figure 3. Schematic of fly anatomy. Picture Credit: Al2 and derivative work by: Giancarlodessi and Georg-Johann (CC BY 3.0).
- Figure 4. Comparison of infested (left) and healthy (right) maggot plants. Picture credit: Joe Ogrodnick.
- Figure 5. Healthy and infested onion plants. Infested plants have a wilted looking appearance. Picture credit: Leonardo Salgado.
- Figure 6. Onion plant infested with maggots. Picture credit: Leonardo Salgado.
- Figure 7. Comparison of healthy and infested maggot plants. Picture credit: Brian Nault.
- Figure 8. Earlier feeding damage by maggots. Picture credit: Brian Nault
- Figure 9. Bulbs infested with maggots. Picture credit: Joe Ogrodnick
- Figure 10. Life cycle of onion maggot (green) and seedcorn maggot (yellow). Picture credits: Joe Ogrodnick.
- Figure 11. Onion seeds treated with different insecticides. Picture credit: Leonardo Salgado.
- Figure 12. Onion maggot fly killed by the entomopathogenic fungus Entomophthora muscae. Picture credit: Joe Ogrodnick.
- Figure 13. Rove beetle that predates maggots. Picture Credit: U. Schmidt, 2022 (CC BY-SA 4.0).
- Figure 14. Comparison of onion plants not treated with insecticides (left side of photo) and plants protected from maggots using insecticide seed treatments (right side of photo). Picture Credit: Brian Nault.
Related Links
- lds223 [at] cornell.edu
- (315) 787-2354
- ban6 [at] cornell.edu
- (315) 787-2379
- mez4 [at] cornell.edu















